Site Navigation :
õ Home
õ Forums
õ Forum FAQ
õ Posting Images on the Forum
õ Photo Competition
õ Links
õ Articles
õ DIY
Administration :
õ
õ Interested in advertising?
Articles Navigation :
õ The Clown Loach
õ Rocks Suitability For Tanks
õ Driftwood/Wood
õ The Nitrogen Cycle
õ The effect
of pH on your fish
õ Hardness
Articles
The following articles are provided for the information of the hobbyist. The information provided here may not be reproduced without the written permission of the webmaster or the original author.
The Clown Loach - Botia macracantha
by Andrea Watts
Index
Introduction
Clown loaches are my favourite aquarium fish. They are colourful, active and have an agreeable nature. These attributes make them an ideal addition to most fresh water aquaria. This article reflects my experiences with clown loaches with particular emphasis on the various patterns that I have encountered. Hope you enjoy my account of these very personable little fish.
Common Name: Clown loach
Scientific name: Botia macracantha
Size (cm) M/F: Up to 30cm both sexes (rarely seen in aquarium examples)
Habitat: They originate from large still and slow-flowing rivers in Borneo, Sumatra and the Sundra Islands. The fin colour will determine their origin; all the ones I have seen are from Borneo. The rivers have a sand substrate, and are quite thickly vegetated and littered with fallen branches. Hence the tank should reflect this arrangement.
Temperature Range: 24C - 27C (Avoid "raw" water at all costs).
Min. Tank Requirements: To get the best out of these guys, I would recommend a tank of at least 250L. As they do best in groups, the larger the tank the better.
Food: Omnivorous, not fussy at all. They love pellets (especially shrimp based), flake, blood worm and most vegetables.
Breeding: Not viable in the home aquarium.
Background
It has been suggested that the name "clown" loach is derived from their striking colour and playful antics. They mostly originate from Borneo and Sumatra, where they live in flowing and still waters. Due to the geographical isolation of these two populations, certain colour differences are a result of bio-diversity. These variations will be discussed later. Kalimantan also has a population of clown loaches although they are not as common in the aquarium trade.
The general colouration, exhibited by these beautiful fish, is bright orange. Arguably, they are the most colourful and best natured loaches available to aquarists. The body is usually marked with 3 broad, wedge-shaped bands of black. The red and black colour on the tail and fins are environmentally determined. Variable colour and markings will be discussed later. Clowns grow to a maximum length of about 30 cm (or 1 foot in old terms). It is rare to see specimens attain this size in aquariums and full grown wild specimens are becoming scarce. The body is somewhat elongated, laterally compressed with an arched spine and relatively flat belly line. The down-turned mouth supports four pairs of small barbels that are situated on the lower jaw. The "lips" are thick and fleshy, thus soft substrate should be used in the aquarium.
Generally speaking they are hardy and long-lived, although they are intolerant of "raw" water. I have kept them in many different water conditions, ranging from quite soft to very hard water. Regular partial water changes are conducive to keeping happy, healthy fish.
Clowns are gregarious by nature i.e. they prefer the company of other clowns. Groups of at least 3 or more is essential to maintaining the well-being of clown loaches. As mentioned in the above overview, they prefer an environment that provides adequate hiding places. They rest upon logs and substrate, quite often in such a manner that it causes alarm for their owner. Many a time I have sprung up from a chair to tap the glass, only to see the offender spring back to life and swim off. This "resting" position is typical of the species. They are known to school with other fish of similar colour and markings; for example tiger barbs. My Distichodus sexfasciatus schools with them all day! "Whiskering" of other fish is common but it appears to have nuisance value only. They spend most of the day schooling together and foraging through the substrate.
Clown loaches enjoy a wide variety of foods. Being mostly carnivorous, they seem to prefer high protein commercial pellets, but relish blood worms. I tend not to add live worms to the tank for fear of introducing parasites. Vegetables are also a welcome dietary addition. I regularly supplement feeding with partially cooked pumpkin, broccoli, zucchini and peas. Fresh lettuce is also greedily accepted. The territorial "clicking" noises exhibited as they feed are quite audible to the human ear. Their particular fondness for snails makes them an ideal biological control alternative to chemical pesticides.
Variations in colour and pattern
As has been discussed already, basic colouration for this species is orange with black bands. The amount and positon of red in the finnage is determined by origin. These variations are often only detectable when fish from different locations are placed side by side for comparison.
Sumatran loaches have relatively intense colouration. The colour contrasts are crisper than those seen in the "duller" fish from Borneo. The most obvious difference between these geographically separated populations is detected in the pelvic fins. Clown loaches from Borneo exhibit a basal and central black area, with a reddish orange leading ray. Sumatran fish have a solid reddish orange fin without any black. Other noted differences include less colouration of the dorsal and anal fin leading edges and a lack of stripe continuation into the caudal peduncle of Sumatran fish. Fish sourced from Kalimantan are similar to those from Sumatra, however the red colouration is more intense. The subtle colour variation between Sumatran and Kalimantan aches could be attributed to habitat. Sumatran loaches live in murky, turbid waters, whereas those from Kalimantan prefer the brown, tannic waters of tributaries. Perhaps the dissolved organic matter of the black water environment helps accentuate the reddish/orange colours in the fish. All fish that I have seen available in Australia appear to be from Borneo stock *edit: I just found a Sumatran!*
The patterns that arise within the black banding on the these loaches are fascinating. I am collecting as many different types that money and source allows me. Interestingly, these spots, shapes and stripes can "shift". By this I mean that they can vary the intensity of black in the boundaries and actually slightly change/move the shape of the markings. Fish often grey out" when fighting over leadership or food. There seem to be 7 basic designs, with variations amongst each group. The following are examples that I currently have in my aquaria:
1. "normal" pattern standard 3 stripes

2. 4 stripe (includes broken stripes and saddle combinations)

3. "jigsaw"

4. second stripe "saddle"/ broken stripe

5. third stripe "saddle"/broken stripe

6. spotted

7. joined stripe

The Trade of Clown Loaches
Clown loaches are harvested from the wild for food and aquarium trade. Approximately 20 million fish are exported annually. Due to their popularity, many small (preferably 2 - 8cm) loaches are captured every year and dispersed world wide. Interestingly, Indonesia has posed a ban on the export of fish larger than 10cm. This policy is hoped to protect breeding adults and make captive breeding by external groups more difficult. Larger specimens are considered to be unsuitable for aquarium life (due to their carnivorous nature and adaptability difficulties) hence large breeding populations are generally left intact. Young fish are only caught during the high water seasons whilst they are abundant and highly concentrated in one area. This makes trapping easier and the economics of such programs are therefore viable during this time. Juveniles adapt easily to prepared foods and captivity.
Collection procedures differ slightly between Borneo and Sumatra. In Sumatra, clowns are caught by using perforated bamboo poles stuck into the river bank substratum. This allows loaches of a preferred size to enter the cavities of the bamboo poles. The poles are set at a pre-determined distance apart, where they are later collected by boat. The contents are emptied directly into the bottom of the boat that is filled with water. The loaches are then taken back to a holding facility where they are sorted according to size and sold to a wholesaler.
In Borneo, a slightly different collection method is implemented. Bamboo poles are bundled and tied together then sunk with stones. Rope is used to attach the trap to stakes that are driven into the river bed. When the trap is retrieved from the river, any loaches that have taken refuge inside the bamboo configuration are shaken into a collection vessel. This shaking action is more detrimental to the well-being of the loaches than the method adopted in Sumatra.
Most fish caught in Kalimantan are used for food and few numbers are exported for aquarium trade.
Although the clown loach trade has heavily exploited numbers in the wild, relatively large populations still exist. This is mainly due to the efforts of the individual governments to control numbers that are caught and the fickle nature of the market.
Although some fisheries in Thailand have capitalised on the artificial breeding of these fish, generally this practice is not viable at this point in time. Before the pressure is taken off wild populations, huge technical advances in the field of aquaculture must be reached. Hopefully this is in the "not too distant future". It would be disastrous to lose this fish from the wild. Environmental disasters (natural and human induced) continually threaten the clown loach in the wild. We don't need fisheries mismanagement
also contributing to their demise.

Credits for this article include:
Loaches Online
Raffles Museum of Biodiversity Research
Rock Suitability For Tanks
By Andrea Watts
Index
Why rocks, aren't rocks!
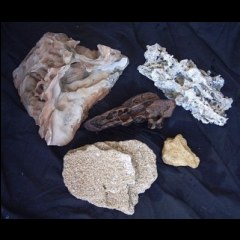
This article serves to provide a brief introduction to rocks and their properties, and hopefully guide you in your choice of decor for various biotope simulations. All text and photos (except fig. 8) are © Andrea Watts. Do not reproduce them without permission of the author
Rock Types:
To make a decision about rock selection for aquarium use, it is beneficial to understand the properties of each rock group. The following is a skeletal lesson in geology that will (hopefully) help you understand why some rocks are less suitable for the use in home aquariums than others. Only the characteristics that affect freshwater environments will be discussed.
Rocks are grouped into 3 major types: igneous, sedimentary and metamorphic. Each of these types consists of subgroups. We will start with igneous rocks.
Igneous Rocks
These rocks are formed from molten material (magma): those which contain high amounts of quartz (SiO2) are termed "acidic"; those without quartz are termed "basic". Rocks with small amounts of quartz are called "intermediate". Common types of acidic rocks include granite (fig. 8), rhyolite, pegmatite and obsidian. Intermediate rocks include syenite, diorite, andesite and trachyte. Those that are termed as basic include gabbro, dolerite and basalt.
Generally speaking, these rocks are suitable for use in the aquarium. However they form sharp edges along broken faces, so care should be taken to smooth off any dangerous projections. Obsidian is glass-like and should be treated with extreme care. Basically, igneous rocks are non-porous, however many contain some minerals that are chemically altered to clay after a period of weathering. With the exception of obsidian, some are then capable of "sponging" up chemicals/toxins from their environment and releasing them slowly into the aquarium. Water worn and polished specimens possess little to no absorption properties, and often appear more realistic in an aquatic environment.
Basic rocks often contain higher amounts of iron-based minerals; however their release is too slow to cause a significant shift in the water chemistry. Igneous rocks do not help buffer water, are generally inert and are non-contributors to providing ideal water chemistry for alkaline dwelling cichlids. They provide decorative properties only.
Sedimentary Rocks
Most rocks chosen by aquarists fall into this category. The main feature that brings this great variety of rocks together is that they are deposited in layers or beds that originally are reasonably flat and well organised. Those that are formed from detrital sediments include sandstone, conglomerate, breccia and shale. Sandstones can vary greatly in their chemical composition.
This is due to the fact that the sand is cemented together by various minerals which have been formed in the pore spaces in the rock. The most common "cements" are calcite, iron or quartz. Sandstones, on the whole, are inert in water, although the cement type will dictate their strength and mineral composition (fig. 1).
The most important factor to consider when utilising these rocks is their porosity and often fragile nature. Sandstones bonded with iron will remain stronger over time than those containing calcite: the latter tending to crumble after a period of submersion.
The high porosity of sandstones leads to the containment and release of toxins/chemicals acquired from an aquatic environment. You need to be sure of their source. Conglomerates and breccias are rocks that are formed
by "clusters" of larger sized particles (fig. 2). They are cemented in a similar fashion to sandstones.
Shales and mudstones are generally not suitable for aquarium use. They are clay based and often fall apart after a period of submersion.
Limestone's are the most commonly used rock group in home aquariums. There are a number of types of limestone, and their properties correspond with the origin of formation (fig. 3.). Some are produced by or from organic material (shells, coral or algae), others originate from chemical activity (oolite and dolomite) and the rest are formed from fragments of calcareous material (clastic). Although
limestone's can be deposited in freshwater, the vast majorities are marine deposits. They are commonly formed in a reasonably clear sea, largely free of mud and sand.
These rocks are usually pale coloured, being grey or even white, the colour
though will depend on the amount of detrital material present. They may be brownish when iron minerals are present and almost black if they contain high levels of mud and organic carbon. The percentage of calcium carbonate present differs between types (chalks containing up to 90%) whereas dolomites contain over 15% magnesium carbonates.
Limestone's are useful for use in hard-water, alkaline biotope simulations such as Malawi and Tanganyikan. They possess good buffering properties, are relatively easy to come by and provide aesthetically pleasing displays with a little imagination. Limestone will often break and form quite sharp fractures. Make sure that you do not leave hazardous projections that may injure your fish.
Evaporatives and ironstones include salt, gypsum, potash ore marl and ironstones.
Only the ironstones are suitable for aquarium use. As suggested, they often contain the following iron-rich minerals: chamosite, limonite, siderite, hematite and magnetite, and are commonly added to planted aquariums to help supplement iron levels. Iron content should be regularly monitored if ironstone is included in your aquarium (fig. 4, fig. 5 and fig. 6).
Metamorphic Rocks
Generally speaking, these rocks are formed from the alteration of igneous and sedimentary rocks through heat and pressure. Examples include hornfels, metaquartzite, marble, slate, phyllite, schist and gneiss. All of these rocks are suitable for use in the aquarium. However, as has already been discussed, their origin will determine the chemical properties that they individually possess.
Of these, marble is the most commonly used metamorphic rock. This rock develops when limestones are intruded by magma, or overrun with lavas. The heat from the molten igneous rock brings about profound changes in the original limestone. Marbles are generally pale coloured rocks of medium to course grain size. The main mineral in marble is calcite; the other metamorphic minerals develop from impurities in the original rock. Due to the thermal alteration of the original limestone, marble is very hard and durable. It is commonly used as a substrate in hard-water Africa aquariums, or as a filter media. It has an excellent buffering capability, and provides a more environmentally friendly alternative to coral products.
In General
Water becomes hard by dissolving soluble salts from the rocks or soil over or through which it flows. Some rocks, for example, slate, granite and gneiss, contain little or no soluble material and have a negligible effect. Others, most notably limestone, are quite the opposite. Hence rocks may affect water chemistry: corals and shells are largely calcium carbonate and some gravels often contain fragments of these substances. Hardness free rocks are a pre-requisite of the soft-water aquarium.
A point not often realised is that some rock surfaces are too rough for use as a spawning substrate by many species. Rocks that would be suitable include granite, schist, gneiss, slate (fig. 7.) and sandstone.
Rock is sold by weight and can be expensive, so you may be tempted to collect your own - but don't do so unless you are able to identify different types of rocks and spot any contaminants in them. It is an offence (by law and on the environment) to collect rocks from the ocean, river courses, bushland and the like. Heavy penalties are incurred for this sort of action.
All rocks must be thoroughly cleaned and scrubbed to remove soil particles and other foreign bodies. Small pieces can be boiled for 10 minutes, but remember to allow them to cool. The amount of rock to be used will depend on the biotope you are trying to replicate, and may range from scattered stones on the floor of a forest stream, to large and complex rock piles representing areas of rapids or a rocky lake shore.
Stones can be used to support raised terraces of substrate material. Never position large rocks on top of the substrate - always bed them in so that substrate slippage or fish excavations cannot undermine them. Always make sure rocky structures are solidly constructed so that they cannot collapse, crushing fishes or crashing through glass; consider sticking them together with silicone sealant for added security and stability.
Never forget that in choosing and arranging rocks you must always keep the fishes' requirements - water chemistry, shelter, swimming space, spawning sites and so on - in mind, and be prepared to forego any ideas which may please your eye, but cause them physical or psychological discomfort. Your aim should be to provide them with a replica of their natural environment in which they will feel at home. They reward you by looking their best.
|
|
|
|
|
|
|
|
|
|
|
Driftwood/Wood For Tanks
By Andrea Watts
Index
The Use Of Wood In Aquariums
Why use wood in your aquarium?
Pieces of cured wood/bogwood provide shelter for shy fish and spawning sites, as well as a visual counter piece for the aquarium. Many plants that possess rhizomes instead of true roots require such structures for adhesion to grow. In addition, many sucker-mouth catfish require wood to rasp upon in order to aid digestion. It is also a viable way to provide the environment and water chemistry required by many soft water (Blackwater biotope) species.
Sourcing the wood
Most local fish shops have pieces of wood for sale that are suitable for use in your aquarium. Some stores may have examples that have been pre-soaked. However the majority of pieces will probably require some degree of preparation before using in you aquarium. Most commonly you will find mangrove roots, eucalyptus species and pre-packed pieces from Africa and Asia (often wrapped in cellophane). The former type will probably contain less tannin than the latter two, however ultimately they are all likely to leach to a certain extent. The pre-packed pieces may have been treated with chemicals as part of the quarantine fumigation process on arrival to Australia. I tend to treat these pieces with suspicion.
Be aware that it is both irresponsible and illegal to remove wood in any form from its natural habitat. Offenders who are caught taking wood in any form incur heavy penalties. Even pieces that lay rotting on the ground provide important habitat sites for a myriad of native animals, and ultimately return nutrients to the soil upon their decomposition. Removal of wood from waterways (lakes, rivers, public dams and oceans) also falls under this category.
However, if you have access to wood from private farmland, or possess a firewood collector's permit, then you may find some suitable pieces. Most eucalyptus species are suitable, however not all natives are safe. Some exude dangerous oils and toxins once submerged, so care needs to be taken. Remember to carefully check the wood for geckos, skinks, spiders, centipedes, scorpions and other small animals before taking it from the site. Carefully remove any bark to reveal hidden organisms.
Wood recovered from riverbanks and waterways may possibly contain significant levels of herbicides, pesticides, salt or phosphate. Due to the porosity of wood fibre, these contaminants are soaked up and then released into the home aquarium. Do not use wood gathered from these locations if you are unsure about the inclusion of these chemicals.
Coconut shells and cork bark are also ideal for soft water situations.
Preparation for Use
Whether you have purchased or collected the wood yourself, it is essential to wash off any residual soil or other foreign particles. High pressured hoses, scrubbing brushes and a screwdriver are useful tools. Remove any moss or lichens, as some will release toxins into your aquarium. Soil often harbours harmful micro-organisms and spores, so you need to be scrupulous in your cleaning regime. Wood of marine origin may have barnacles or mussels attached: they should be removed if they pose a threat to the inhabitants of the tank. Coconut shells should be free of fibre and nut residue.
These wooden items can be varnished for use in alkaline conditions, but check that none of your fishes has a taste for nibbling wood (e.g. Ancistrus spp.), as many varnishes are toxic if consumed. Wooden items have a disconcerting tendency to float until thoroughly waterlogged, and may need to be wedged into place with stones.
There is also an increasing range of simulations of wood, including resin and fibreglass replicas. These range from the remarkably realistic to the truly awful; fishes don't appear to be aesthetically affected, so the choice is up to you.
Tannin: description and removal
Tannin (generally short for tannic acid) is an organic, dark substance that occurs naturally in most plant material. It is generally harmless to most fish. Rainforest biotope simulations benefit from the use of bogwood, coconut shells and cork bark. All of these will tend to leach tannins into the water, acidifying it and turning it brown; even in acid-water aquariums this can be too much of a good thing. Hence new materials of this kind should be "aged" by soaking or leaving them outside to weather.
The amount of tannin that a piece of wood contains is dependent on:
1. The species of tree from which it came;
2. The size/density of the piece;
3. The period of time which it has been separated from the tree (period of weathering); and
4. The source from which it is acquired (aquatic environment vs. terrestrial).
The following techniques are used to reduce the level of tannin that is leached:
1. Small pieces can be boiled for 2 hours (the addition of rock salt can hasten this process);
2. Larger pieces can be weighted down and submersed in an old tub for a period of time. The water needs to be changed regularly to dilute the escaped tannins in the water;
3. The use of activated carbon in the filter will greatly reduce the visible coloration;
4. Purigen, a Seachem product, is highly effective in the removal of minute organic particles in the aquarium; and 5. Simply leaving it in outside to weather naturally.
It must be said that some pieces of wood will continue to leach tannin into the aquarium for many years after their addition. In this case, regular water changes will help to prevent the colour from becoming unsightly.
The Nitrogen Cycle
by Anthony Kendrick (4th year BEd Student)
Index
This life nurturing cycle is a must know aspect for all forms of fish keeping. As a guide for beginners this article has been developed so that they can become aware of its importance in sustaining the life of an aquatic ecosystem. The article will focus on the basics of the nitrogen cycle rather than getting into the nitty gritty of how the cycle works. After firstly describing the cycle, connections will be made as to the general care of an aquarium and ways in which you can nurture your delicate ecosystem.
The focusing questions will be:
What is biological filtration? What is mechanical filtration?
Which filtration should you use (the different options available)?
How much water should your filter be filtering?
How can I test the water for impurities?
The information provided within the article will be collected from various websites and from opinions of people in the industry and experienced hobbyists. In no circumstance do I claim to be an expert but I will use my experience within the hobby and my skills developed as a pre-service teacher to research and explain the nitrogen cycle so as it can be easily understood by beginners.
Chemicals involved in the nitrogen cycle
Nitrogen:
Nitrogen (N) is an odourless, colourless gas. This gas represents approximately 78% of the atmosphere. It is a vital element that is essential to living ecosystems. It is a plant nutrient that can cause an abundance of bacteria and algae when high amounts are present. In water it occurs in various forms. These forms are nitrite, nitrate and ammonia.
Ammonia:
Ammonia (NH3) is the first form of nitrogen released when organic matter decays. This chemical compound is a highly toxic chemical to aquarium fish and is probably the biggest killer of fish. The most common way it occurs is by over feeding your fish.
Nitrite:
Nitrite (N02) is a toxic chemical created within the second step of the nitrogen cycle. It is easily removed with biological filtration and should be at 0 within your aquarium.
Nitrate:
Nitrate (NO3) is a chemical compound containing oxygen and nitrogen that can exist in the atmosphere or in water it can have harmful effect on humans and animals at high amounts. It occurs in the final stage of the nitrogen cycle. Low amounts are not harmful in your aquarium.
The Cycle
The nitrogen cycle describes how organic wastes break down in the aquarium. The whole process begins with the introduction of organic waste within your aquarium. This can come from uneaten food, excrement, decaying plant life or it is present in your water when added to the aquarium.
During decomposition ammonia is released into the aquarium water. Within a healthy aquarium there is an abundance of bacteria that is often referred to as good bacteria (technical term nitrosomonas bacteria). This good bacteria converts ammonia into nitrite. At this stage the chemical has gone from one form of toxic form to another.
Another form of good bacteria present within a healthy aquarium is a Nitrobacter bacterium. This breaks down the toxic nitrite into a less toxic form of nitrogen called nitrate. Throughout this process the forms of good bacteria multiply which allows a greater capacity for ammonia to be broken down into a non toxic form.
Good bacteria are harboured within your filter, in the flowing water, attached to gravel, tank ornaments and plants.
Organic Waste ----------> Ammonia ----------> Nitrite ---------->
Nitrate ----------> *Water Change
*Water change removes a percentage of nitrates
The new aquarium
When you start a new a new aquarium the aquarium and filter is sterile. Therefore if you introduce fish into your tank straight away you will get an initial ammonia spike at around 1 week. This is likely to kill your fish. This is known as new tank syndrome. This occurs because there is no or insufficient amount of good bacteria to break down the toxic ammonia. The way to avoid the death of your fish is to cycle the aquarium before you introduce your fish.
Cycling the new aquarium.
There are 2 common methods of cycling your aquarium. One method is called fishless cycling and the another method is used with the presence of fish.
Fishless cycling using live bacteria cultures:
This method uses either live bacteria purchased from a shop or squeezed out of a sponge from an established filter. The trick is to put in a small amount of food every day to produce ammonia spikes. The small amount of live good bacteria will begin to de-toxify small amounts of ammonia and allow the good bacteria to reproduce. Within 3 to 4 weeks there should be sufficient amounts of good bacteria to sustain the
aquatic ecosystem, at this point fish can be introduced.
Cycling the aquarium using gold fish:
Gold fish are somewhat resistant to ammonia to a certain point. Therefore you can use them to cycle your aquarium. Organic matter will be introduced into the
ecosystem through excrement and uneaten food. As this matter decays ammonia will be produced and the process will continue as mentioned above.
Ammonia Spikes and Nitrite Spikes:
A spike will also occur in a healthy tank environment if the tank isnít properly maintained. Over amounts of good bacteria can cause spikes. This emphasises the need to change a certain percentage (30-40% a fortnight) of your aquarium water regularly and cleaning your.
Over feeding your fish will also result in a spike due to an over amount of organic waste material. Care must be taken when you do water changes and cleaning your filter. You must use water that has been treated with water ager to get rid of the chlorine and chloramines, which are chemicals that will kill all off the good bacteria within your tank or filter media. Water ager can be found at all local aquarium or pet stores.
In order to monitor and prevent fish loss due to ammonia, nitrite or nitrate spikes it is important to test for the presence of these chemicals within your aquarium. Test kits can also be found at most local aquarium or pet stores. If you find any levels of ammonia or nitrite (or a high level of nitrate) within your aquarium you may need to perform a water change (30%). This will allow Nitrate levels to be diluted.
Filtration
As mentioned earlier, your filter harbours good bacteria within your aquarium. As the water flows through your filter it will do both of the following.
It will filter the organic particles through a sponge or filter cloth (mechanical filtration) or
the good bacteria attached to filter medium will biologically detoxify any toxic materials within your tank (biological filtration).
A power filter (or sponge) will do a small amount of biological filtration but will do mostly mechanical filtration. This method is ok to use in tanks up to 250 litres in my opinion. However biological filtration is much more efficient in running a healthy aquarium as it harbours more good bacteria. Biological filtration can be seen in trickle filter systems and canister filter systems. These filtration systems also allow particle filtration. These systems allow greater surface area for the good bacteria to attach to therefore the tank has a greater capacity to detoxify the water. With any tank size it is important that you know how much water your filter is filtering per hour. On any given tank the water should be filtered at least 4 times an hour. Every time the water is passed through the filter it is not entirely detoxified, therefore it needs to go through the filter system several times to keep the water safe for youíre your fish. If a tank is 250 litres it must be filtering at least 1000 litres per hour.
This process is also true for back yard ponds.
The effect of pH on your fish
How do I acclimatise the fish to these adjustments?
by Cichlids_au
Index
When adjusting pH, you need to raise it slightly
each day until the pH you require is achieved
To be precise you need to raise the pH by not more that .03pH units per day.
Remember that the pH scale is logarithmic, so a one unit shift = a ten fold
change in hydrogen ions. So a small pH shift indicates a substantial change.
Fish, like us, are just complex semi permeable membranes; they need to keep a
constant internal pH level and an acid/alkali (or acid/base) balance in their
blood. They use basic bicarbonate ions and or acidic carbon dioxide to do this.
If the blood becomes too acidic because thereís to much carbon dioxide as a
result of metabolic waste the blood plasma level of bicarbonate ions rises to
buffer the pH level of the blood back up to normal. The other way is they will
add carbon dioxide or remove bicarbonate ions to help lower the blood pH.
This is speed up by a hormone called carbonic anhydrase in the blood and gill
membranes.
If you change it fast, you can cause your fish to start gasping, the reduced
oxygen carrying ability of the haemoglobin at the irritated gills causes this,
which inhibits gaseous exchange. This is call acidosis. The opposite is
alkalosis and destroys the gill and fin tissue.
Now. When you change your pH, you change the hardness and salinity. Experienced
fish keepers know that to get a perfect and stable pH you should be measuring
your hardness, when itís set, your pH will not vary. It is measured in units
of degrees of hardness dH or Mg/litre Caco3. Salinity is measured by passing an
electric current through the water.
When you change the hardness to fast it affects osmoregulation and causes
osmotic stress, so if bicarbonate ions exceed normal levels to quickly the
bicarbonate excretion from the gills stops, which causes the pH of the blood to
shift causing alkalosis.
When you change the salinity to fast it also affects osmoregulation and changes
the osmotic pressure and the ability to transfer fluids through the semi
permeable membranes (the cells) of the blood and gills is altered. A
physiological change has now occurred to the cells and the fish goes into
stress.
So now you know how you avoid killing your fish by raising the pH to quickly.
Mark Coleman
Understanding and Controlling Hardness
by Cichlids_au
Index
Hardness is a
measure of metallic ions in the water, mainly calcium and magnesium. Calcium
being the most important, because it has up to ten times more ions than
magnesium. The ions are found in three forms, hydroxides, bicarbonates and
carbonates.
Total hardness (GH) is the total content of salts in the water. This can be
divided into two general groups, temporary hardness and permanent hardness.
Temporary can be boiled off but permanent canít. Temporary hardness is
carbonate hardness (KH) and is the major proportion of GH.
Alkalinity (pH) and hardness are interlinked, and alkalinity closely reflects
temporary hardness. Temporary hardness is caused mostly by bicarbonate ions,
which are mostly what causes the alkalinity. Permanent hardness donít
effect it .
Tuck this away in your mind
You can get bicarbonate from the supermarket (500gms for $0.95 at the
supermarket sold as common baking soda. Or 200gms at the aquarium shop sold as
ìpH Upî for $10).
Magnesium (Epsom salts) from the chemist
And that shellís, shell grit and coral are nearly all calcium, as are some
rocks.
To make water harder, add calcium based salts to the water add calciferous
material to the substrate and filter and away you go. That means put some salt
and sodium bicarbonate in the water, (these fluctuate) and shells or shell grit
to the filter or substrate, these will buffer the water and stop the bicarbonate
fluctuations.
Sodium Bicarbonate will raise your pH to 8.0. You can put a tablespoon full in a
glass of water and test it and get 8. You can then fill the glass with sodium
bicarbonate and put enough water in to make it runny, test that and still just
get a pH of 8, but remember that in the tank without the shells, the next day
your pH will be down again.
I donít really know much about making water softer because Sydney tap water is
good for American cichlids and African Riverine cichlids. So I only up the
hardness and pH to accommodate my Rift Lake Africans. To lower your pH you need
to use sodium biphosphate. Or you can use CO2, delivered by an injection
system.
ìpH Downî replaces bicarbonate ions with hydrogen ions, which means
you would need heaps of aeration to break down the carbonic acid the process
would produce.
You can also mix RO water with tap water or you could use rain water if you
lived in an area with plenty of lichen and a clean collection point.
If you needed to reduce the pH and hardness for an emergency you just need to do
water changes with tap water. (In Sydney that is, test your local supply)
Mark Coleman